Customer-Specific Subracks for Anesthesia Systems / Dräger Medical
/in Useful Information /by admin
To meet today’s rigorous client demands, standard 19-in. subracks must be modular, flexible, affordable, and mainly, ready to be utilized in numerous application areas.
However, in some instances it may be more reasonable to construct and individually configure a subrack than using a standard one.
The integration of electronic components into complex medical equipment is an example where specialised customer requirements for location and application might require standard product modifications.
One Platform for All anaesthesia Technologies
Dräger Medical conductor & Co is a world-leading manufacturer of medical equipment that provides integrated system solutions and services in line with the patient process chain in CareArea™, emergency medicine, OP and anesthesia, intensive care, prenatal medicine, and home care.
Dräger Medical’s innovative ideas are the result of years of experience, significantly with the anaesthesia system referred to as Zeus®.
Through data connection and also the integration with hemodynamic monitoring (and, integration with hospital data systems), a healthcare professional can get comprehensive information about a patient’s status with Zeus.
The Zeus anaesthesia system integrates all kinds of anaesthesia, patient monitoring and documentation. Besides manually directed gas dosage, for example, automatic administration of oxygen, carrier gas and volatile anaesthetics can be carried out.
Control Unit: The Heart of the System
A core part of the system’s electronics—the central control unit of the anaesthesia workstation, consisting of several electronics modules — is manufactured by MSC Freiburg GmbH, a developer and manufacturer of electronic components, systems and equipment based in Freiburg, Germany.
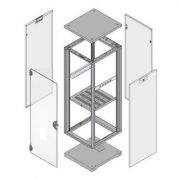
Corresponding subracks were required for housing MSC’s modules. During the design phase, it became clear that a standard 19-in. subrack, available on the market from different manufacturers, could not be used.
Acording to Dräger Medica, even modular and flexibly assembled products couldn’t do justice to the degree of individuality required, which adds that the specified installation areas and fixing possibilities of the end-product made unique construction necessary.
The modular construction was one of theprerequisites determined by Dräger Medical so that components could be exchanged quickly if necessary. In addition, all electronic components manufactured by MSC are shielded from one another by using individual shielding screens to achieve solid electrical safety levels.
Close Cooperation and experience Bring Results
The entire mechanical construction was developed in close cooperation between Dräger Medical, MSC and Schroff, in order to utilize each firm’s design expertice and technical manufacturing skills.
Schroff supplies a pre-assembled 6 U high subrack, with a separated interior for four 3 U plug-in units and a 6 U plug-in unit.
This layout is a standard design capability in EuropacPRO subrack.
MSC relied on Schroff for the know-how and years of experience of electronic design packaging for the development and manufacture of this subrack.
Despite Schroff‘s very flexible 19-in. subrack vary EuropacPRO, only a couple of standard parts like horizontal rails and guide rails couldbe utilised from its portfolio of products.
According to Dräger Medical, about 80th of the subrack had to be constructed from customized components.
The subrack is constructed of aluminum zinc sheet metal and incorporates Schroff subrack components for PCB and backplane integration.
EMC requirements, which are a must for applications in medical technology, were achieved simply because the frame for the assembly into the end-unit consists of 1 continuous part. The front panels from Schroff are equipped with textile EMC seals maintaining EMC integrity standards.
The backplane was integrated at MSC where the module containing the electronic parts was thoroughly tested.
5 modules are installed in each unit, but additional scalability is built into the design.
NEW: Guide Column with Integrated Hydraulic Pump
/in Useful Information /by adminThanks to our guide columns with an integrated hydraulic pump, you will receive a system which allows a simple assembly at your application. As the guide columns are very stable, it is possible to use only one.
Because of the integrated hydraulic pump, you can operate completely without electricity and you can adjust weights up to 10kN (2248 lb) by simply pumping. Strokes can be chosen flexibly between 80 and 400mm.
| Guide column | Pump | Stroke | min. extended length (EL3) |
Force | Model | Index No.* |
| FSC | X | 300 | 710 | – | – | 001* |

|
X = Hydraulic pump | 80 – 400 | 2x stroke + 150 | up to 10 kN | – =Standard V=reinforced B=special |
*only necessary for repeating orders |
| Guide column | Pump | Stroke | min. extended length (EL3) |
Force | Model | Index No.* |
| FSM | X | 250 | 510 | – | – | 001* |

|
X = Hydraulic pump | 80 – 400 | 2x stroke + 150 | up to 10 kN | -=standard B=special |
*only necessary for repeating orders |